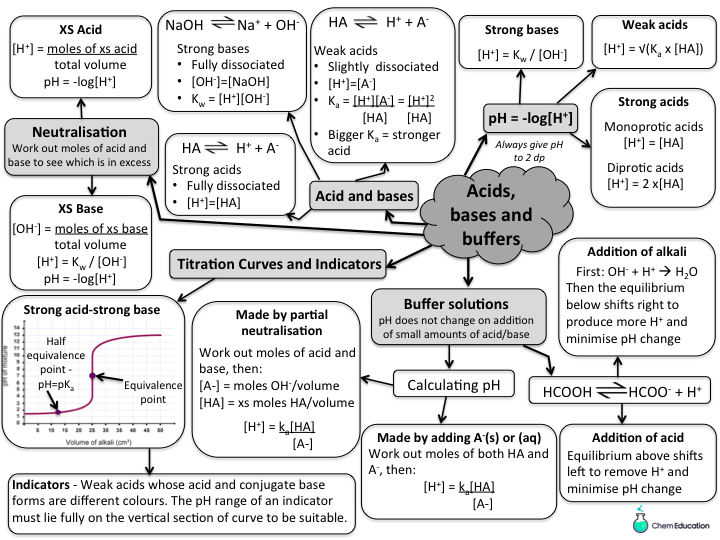
Differentiate The Functional Roles Of Acids Bases And Buffers - An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids.
If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
If a slight decrease below 7.35 in ph of blood, this will act as a. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. A ____can neutralize small amounts of acids or bases in body fluids.
Acids ,bases and buffers D.Pharmacy PPT
An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids.
Acids bases ph and buffers report sheet
A ____can neutralize small amounts of acids or bases in body fluids. If a slight decrease below 7.35 in ph of blood, this will act as a. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
Acids, Bases and Buffers PDF Buffer Solution Acid
An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids.
ALevel Acids bases and buffers Teaching Resources
If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
Solved Acids, Bases, pH, and Buffers Report Sheet Acids,
A ____can neutralize small amounts of acids or bases in body fluids. If a slight decrease below 7.35 in ph of blood, this will act as a. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
Acids, bases and buffers revision bundle Teaching Resources
An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids.
Chapter No 1 Acids, Bases and Buffers PDF
If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
Acids bases ph and buffers report sheet
An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when. If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids.
Acids, Bases, PH, Buffers
If a slight decrease below 7.35 in ph of blood, this will act as a. A ____can neutralize small amounts of acids or bases in body fluids. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
SOLVED Describe the structure and function of water and its role in
A ____can neutralize small amounts of acids or bases in body fluids. If a slight decrease below 7.35 in ph of blood, this will act as a. An acid is any substance that when added to, an aqueous solution increases the [h +] of the solution and a base is any substance that when.
An Acid Is Any Substance That When Added To, An Aqueous Solution Increases The [H +] Of The Solution And A Base Is Any Substance That When.
A ____can neutralize small amounts of acids or bases in body fluids. If a slight decrease below 7.35 in ph of blood, this will act as a.